|
|
Our major research interest is to elucidate the etiologic agents isolated
from pathogenic bacteria related to the worldwide emerging and re‐emerging
diseases.
Studies on the cellular and molecular mechanisms of diarrhea induced by
bacterial enterotoxins:
Aeromonas sobria hemolysin is important in the pathogenesis of diarrhea
caused by this enteropathogenic bacterium. By immunoprecipitation analysis
using hemolysin and anti‐hemolysin antibody, a 66‐kDa protein (p66) was identified
as a receptor for A. sobria hemolysin on Intestine 407 cells. The p66 was
found to be a glycosylphosphatidylinositol‐anchored glycoprotein expressed
on the surface of Intestine 407 cells. (Ref. Microb. Pathog. (1999)27:215)
Focusing on the molecular mechanisms of the diarrhea induced by heat‐stable
enterotoxins (STd) of enteropathogenic bacteria, we are also studying 1)interaction
of Escherichia coli heat‐stable enterotoxin with its receptor and 2)activation
of guanylate cyclase(GC‐C)by STa.(Ref. Eur. J. Biochem.(1999)263:338)
Studies on the pathogenesis of Helicobacter pylori:
To investigate a potential mechanism of how H. pylori establishes infection,
we investigates the host‐parasite relationships of H. pylori, focusing on
vacuolating cytotoxin A (VacA)and Cag pathogenicity island (Cag PAI).
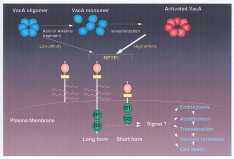
1)VacA exposed to alkaline or acid conditions, with subsequent neutralization,
exhibits enhanced vacuolating activity;the acid or alkali‐activated VacA appears
to bind a cell surface receptor protein of ~250kDa. N‐terminal and internal
amino acid sequence is consistent with the hypothesis that p250 is RPTPβ.
Phorbolmyristate (PMA, TPA) induces differentiation of the human leukemic
cell line HL‐60 into cells with macrophage‐like characteristics and enhances
the susceptibility of HL‐60 cells to VacA. PMA induced expression of RPTPβmRNA
and protein as determined by RT‐PCR and indirect immunofluorescence studies.
Vitamin D3 and IFN‐γ, which stimulate defferentiation of HL‐60 cells into
a monocyte‐like cells, also induced VacA sensitivity and expression of RPTPβ
mRNA, whereas 1.2% DMSO and retinoic acid, which stimulated the maturation
of HL‐60 into granulocyte‐like cells did not. RPTPβ anti‐sense oligonucleotide
inhibited induction of VacA sensitivity and expression of RPTPβ. Double immunostaining
studies also indicated that newly expressed RPTPβ colocalized with VacA in
PMA‐treated HL‐60 cells. BKN‐21 cells transfected with the RPTPβ cDNA acquired
VacA sensitivity. All data are consistent with the conclusion that acquisition
of VacA sensitivity by PMA‐treated HL‐60 cells results from induction of RPTPβ,
a protein that function as the VacA receptor. (Ref. J. Biol. Chem.(1999)274:36693,J.
Biol.Chem.(2000)275:15200)
2)Human β‐defensin‐2(hBD‐2)is an antimicrobial peptide which belongs
to one of the most important host defence system against bacterial infection
in several epithelial tissues. We studied the effect of H. pylori on the expression
of hBD‐2 mRNA in MKE45 gastric mucosal cells. H. pylori, but not culture filtrate,
increased hBD‐2mRNAlevel in MKN45 cells, whereas thus inductive effect of
H. pylori was not detected when Intestine407 cells were incubated with H.
pylori. Among the tested strains of H. pylori, which lacks Cag PAI, did not
induce hBD‐2 mRNA in MKN45 cells. These results suggested that cag PAI of
H. pylori is important for inductive expression of hBD‐2 mRNA in MKN45 cells.
Exposure of MKN45 cells to Salmonella typhimurium, S. enteritidis, S. typhi,
and S. dublin, but not Escherichia coli ML35, resulted in remarkable induction
of hBD‐2 mRNA.(Ref. Biochem. Biophys. Res. Commun.(1999)283:770,Infect.Immun.(2000)68:1806)
Studies on the development of cholera vaccine.
The overexpression of fimbriae of Vibrio cholerae O1 is uuder study for
use in cholera vaccine trial. (Ref. Microbiol. Immunol.(2000)44:439)
| Professor | Toshiya Hirayama |
| Assistant professor | Yoshio Ichinose |
| Research Associate | Masahiko Ehara< |
| Research Associate | Akihiro Wada |
| Research Associate | Akitoyo Ichinose |
| Technologist | Mamoru Iwami |
| Technician | Kayo Honda |
| Postgraduate Student | Takahiro Kimura |

Cytotoxicity of VacA toxin through its binding to receptor‐protein tyrosine phosphataseβ

Laboratory